
However, Dyke says that replacing the Bohr model in his curriculum would be difficult, and not just because it’s easier to learn. After school, people tend to remember the image that they spent more time with. 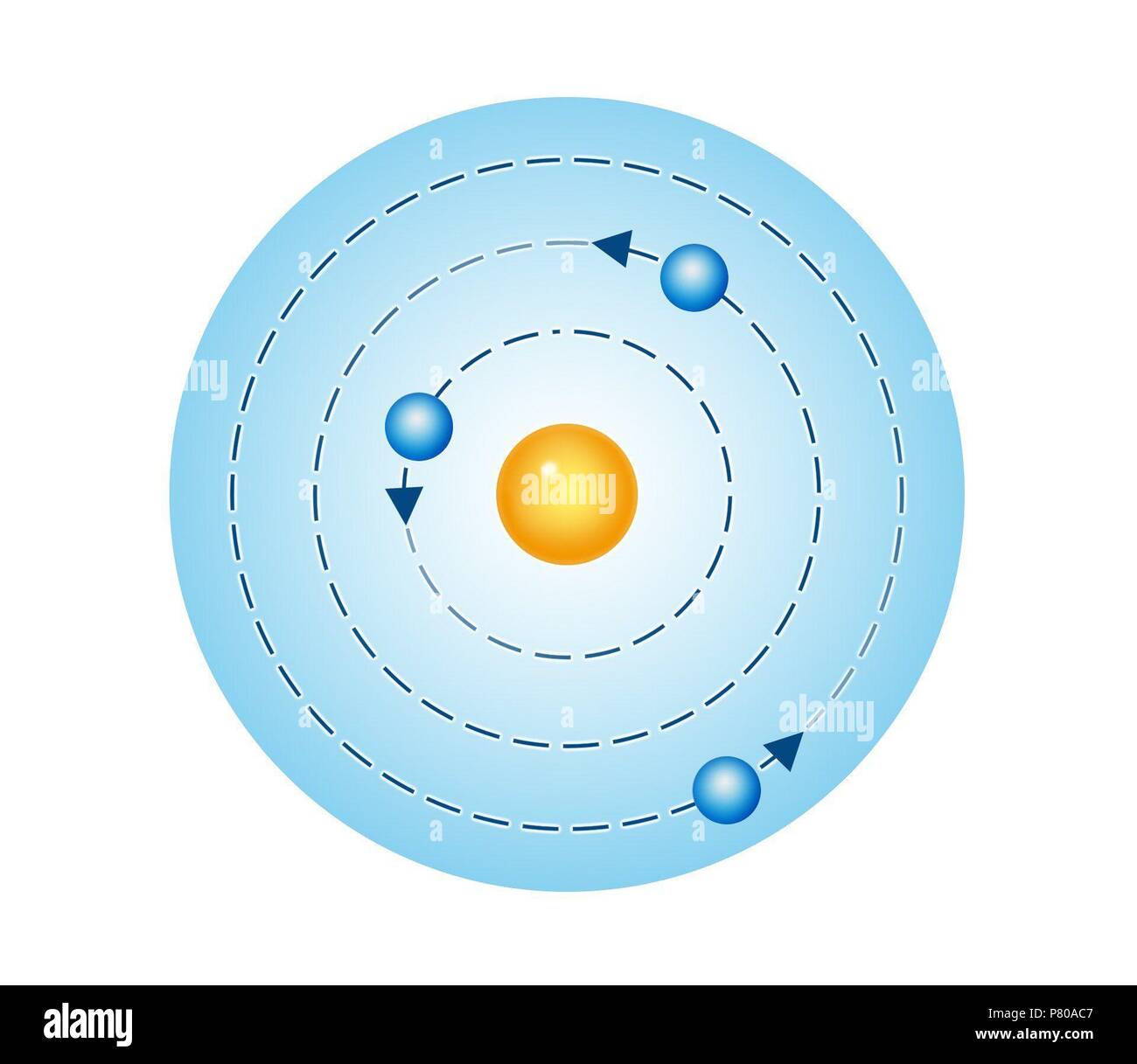
When it comes to the more modern models, Dyke says “we tend to only touch on those briefly and leave it behind” because there just isn’t the time. This is tricky material, and Bohr’s picture of the atom is simple and works well enough, so it’s a good introduction. So why has the Bohr atom stayed around? “It gives us a good place to start the conversation about the composition of the atom,” says high school chemistry teacher Dr. Credit: Public Domain via Wikimedia Commons Nearly no results show the strange, balloon-like shapes that have accurately described the deepest workings of chemistry for nearly a century.Ī model of the atom based on quantum mechanics. Image search for “the atom” online, and the vast majority of results show his atomic solar system. It explains the lives of atoms much better than Bohr’s model does, and science has never turned back.Īnd yet it’s Bohr’s model of the atom that people recognize most today. The new model, called quantum mechanics, took a page from Bohr but took it to a whole new level. Just four years after the Nobel Prize, a huge sea change in physics made Bohr’s model obsolete. Unfortunately, not all of these issues could be fixed. Another scientist came up with the idea that two electrons could share an orbit, an idea that ended up outliving the Bohr model itself. One scientist got better results when he tried oval-shaped orbits, which quickly became the norm. Bohr and others spent the 1910’s and 1920’s debating how to make the model better. 
It got hydrogen right, but it failed miserably at describing the other elements. The Norwegian Nobel Committee moved fast (for them!) to award Bohr the prize in physics in 1922.īut all that success doesn’t mean the model was perfect. Not everyone had been on board with Rutherford’s model, but Bohr’s hard work cemented its place in science. In other words, he’d taken that sketch of a bike and painstakingly drawn in working gears. Among its successes, Bohr’s model of hydrogen explained why hydrogen lamps (picture neon signs) glow red instead of another color. Amazingly, it only took Bohr a year to breathe life into his boss’s model and make it his own. Only a few years earlier, Rutherford had discovered roughly how an atom was built, but his model couldn’t explain how it worked, much like a sketch of a bike without its gears. In 1912, he fatefully took on his mentor’s old project: the structure of the atom. When that relationship quickly soured, he simply arranged to work with another Nobel laureate by the name of Ernest Rutherford. He landed a job with a Nobel Prize-winning professor. By the time he earned his Ph.D., Bohr was a force to be reckoned with. In college, he worked obsessively in a competition meant for mature scientists and won its gold medal. Physics was changing fast at the start of the 20 th century, when a young Niels Bohr was out to prove his worth. You can find it on countless science t-shirts and in TV shows like The Big Bang Theory, but here’s a reality check: scientists replaced it way back in the 1920’s.īut before we start pushing for more accuracy in our media (and I think we should), this old model still has something to teach us about how scientists today tackle big problems. It pictured electrons orbiting a central nucleus like planets orbit the sun, and it’s still the most common picture of the atom today. Way back in 1913, everyone hailed Niels Bohr’s new model of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
